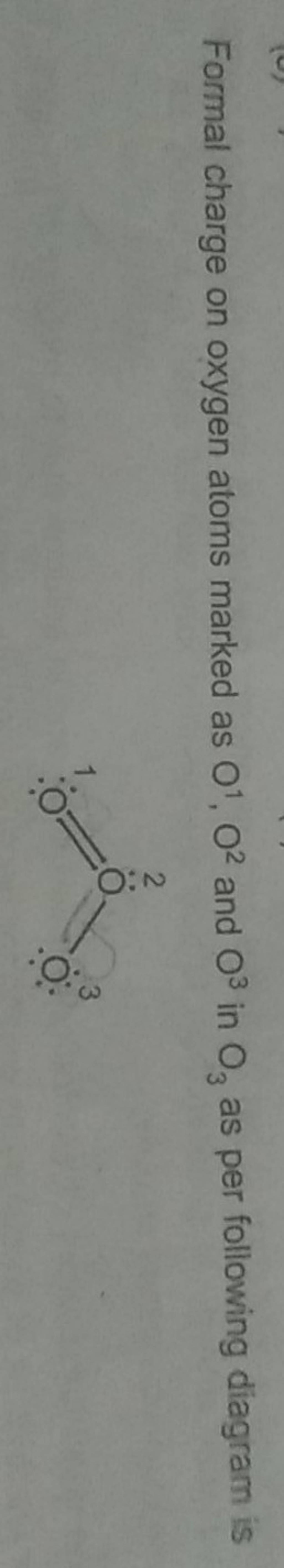
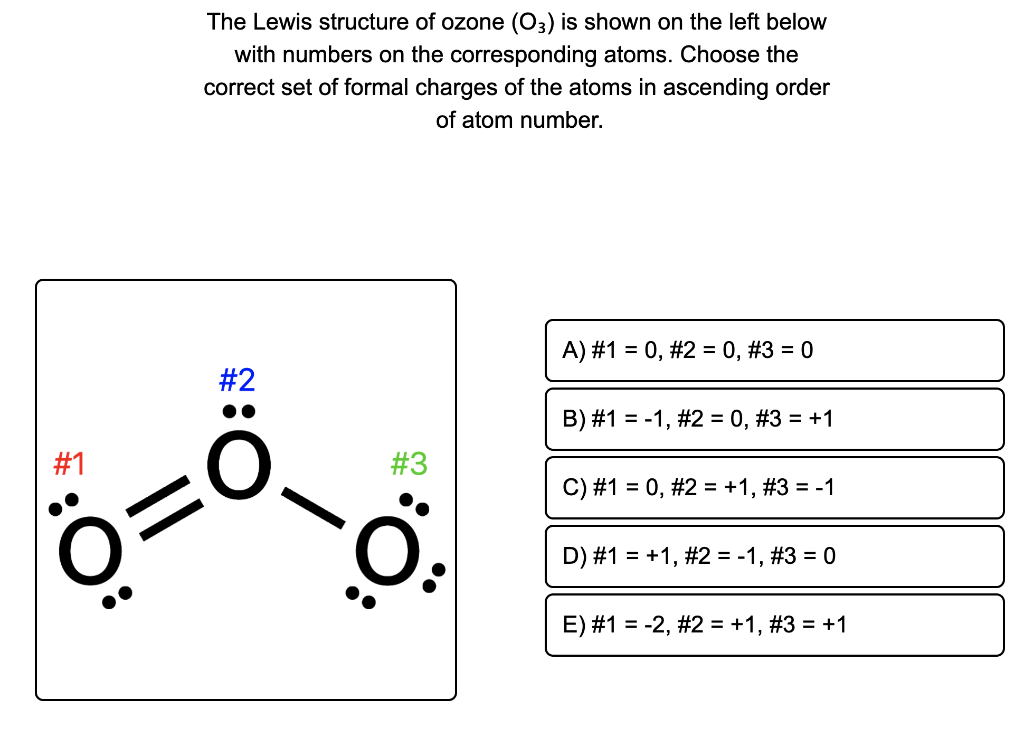
Formal charge on oxygen atoms marked as O1,O2 and O3 in O3 as per follow..
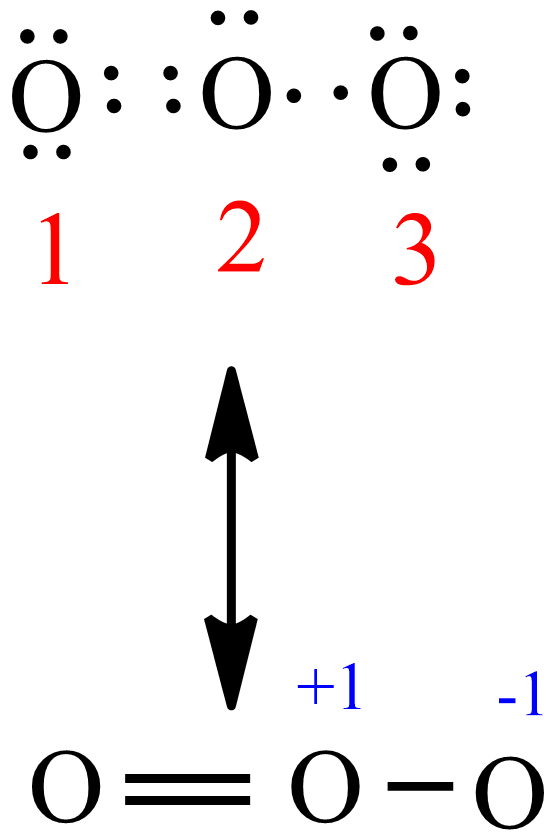
The formal charge on O: 6-2-½ (6)= 1, thus the formal charge on o3 Lewis structure is +1 on the central oxygen atom. Similarly, two adjacent oxygen atoms carry (-½ ) partial negative charge, and central oxygen carries +1 formal charge as shown in the figure below. O3 lewis structure formal charges. By applying this formula, we find that each.

How to Calculate the Formal charge of O3 YouTube
In order to calculate the formal charges for O3 we'll use the equationFormal charge = [# of valence electrons] - [nonbonding val electrons] - [bonding electr.

Formal charge on oxygen atoms marked as O1,O2 and O3 in O3 as per follow..
Formal charge in O 3 ( Ozone): In an O 3 molecule, the formal charge on the middle oxygen atom ( 2) is + 1. In an O 3 molecule, the formal charge on the left oxygen atom ( 3) is - 1. In an O 3 molecule, the formal charge on the right oxygen atom ( 1) is 0.

Calculate formal charge of central atom O3 O=[Os] Filo
Explanation: Simple VESPER requires that we distribute 3 ×6 = 18 valence electrons across 3 centres: O = O+ − O−. From the left, O1, has TWO lone pairs; O2 has ONE lone pairs; and O3 has THREE lone pairs. And thus the formal charge of each oxygen atom ( 8e−,7e−,9e−) is 0, + 1, −1 respectively.
no2 bond order
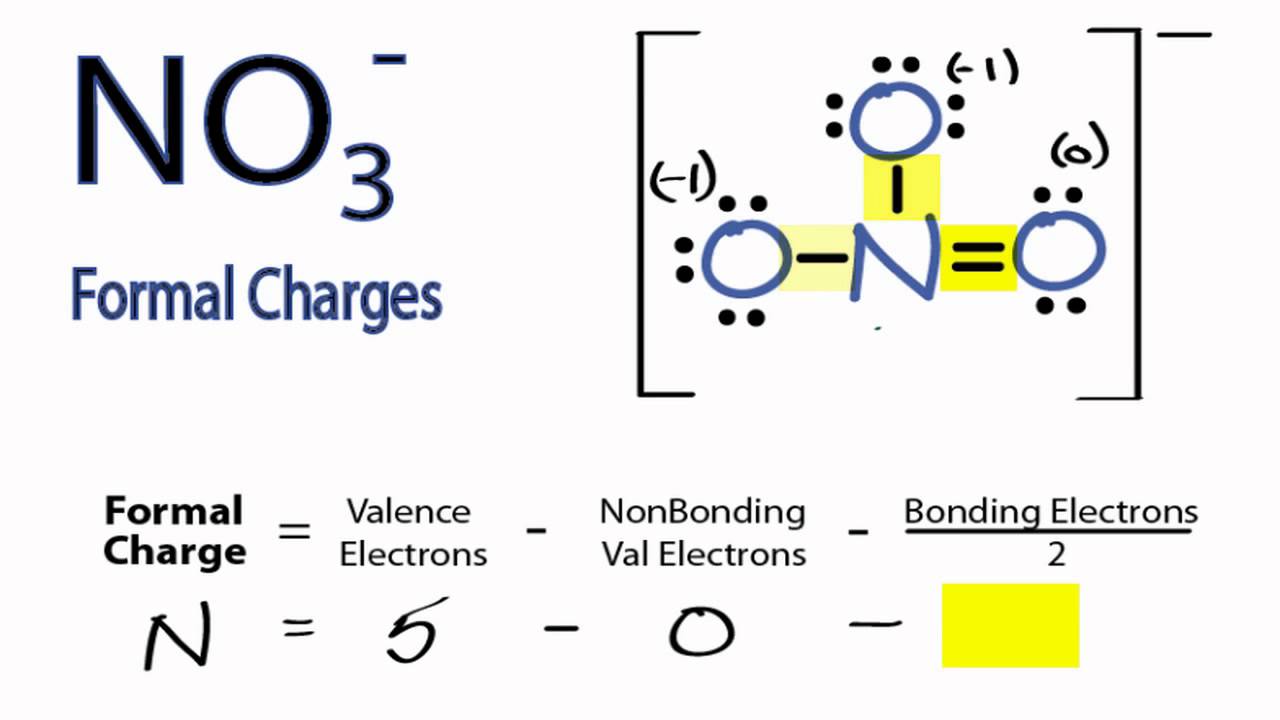
The correct option is B 1. Formal charge (F C) = V −L− B 2. Where, V = Total number of valence electrons in the atom. L = Total number of non bonding (lone pair) electrons in the atom. B = Total number of bonding (shared) electrons in that particular atom. Hence, the formal charge on the central O atom in O3 = 6−2− 1 2×6= +1. Suggest.
FORMAL CHARGE
How to calculate the formal charges on the atoms of ozone (O3)MOC members get access to over 1500 quizzes on O3 and many other topics, plus Flashcards, the R.
What are the formal charges in "O"_3 (ozone)? Socratic
In the O3 Lewis structure, there is one double bond and one single bond around the oxygen atom, with two other oxygen atoms attached to it. The oxygen atom. Formal charge = valence electrons - nonbonding electrons - ½ bonding electrons. For left oxygen and right oxygen atom, formal charge = 6 - 6 - ½ (2) = -1.
Solved The Lewis Structure Of Ozone (03) Is Shown On The
> P block elements Class 12 > O3 Lewis Structure Formal Charge O3 Lewis Structure Formal Charge. O3 Lewis Structure Formal Charge. February 23, 2023

Calculating Formal Charge of O3 A StepbyStep Guide Actualizado October 2023
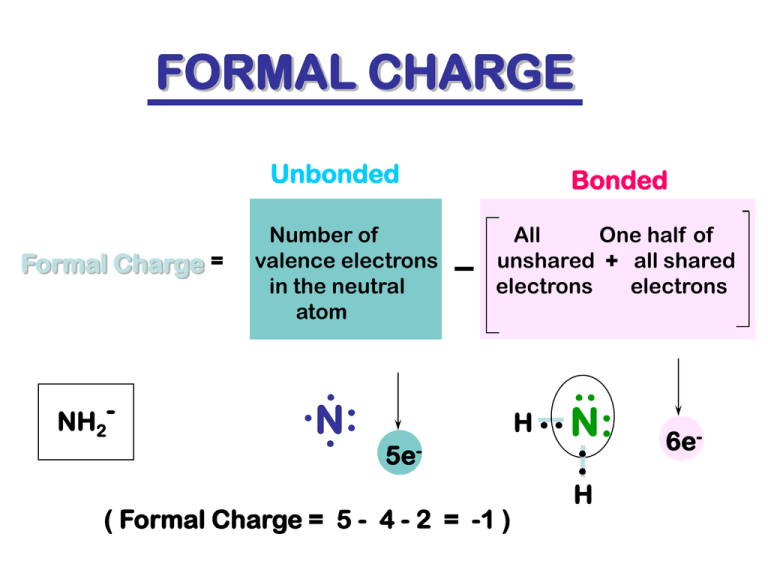
The formal charge of any atom in a molecule can be calculated by the following equation: FC = V − N − B 2 (1) (1) F C = V − N − B 2. where V is the number of valence electrons of the neutral atom in isolation (in its ground state); N is the number of non-bonding valence electrons on this atom in the molecule; and B is the total number.
Find the formal charge of ‘O’ in ozone.
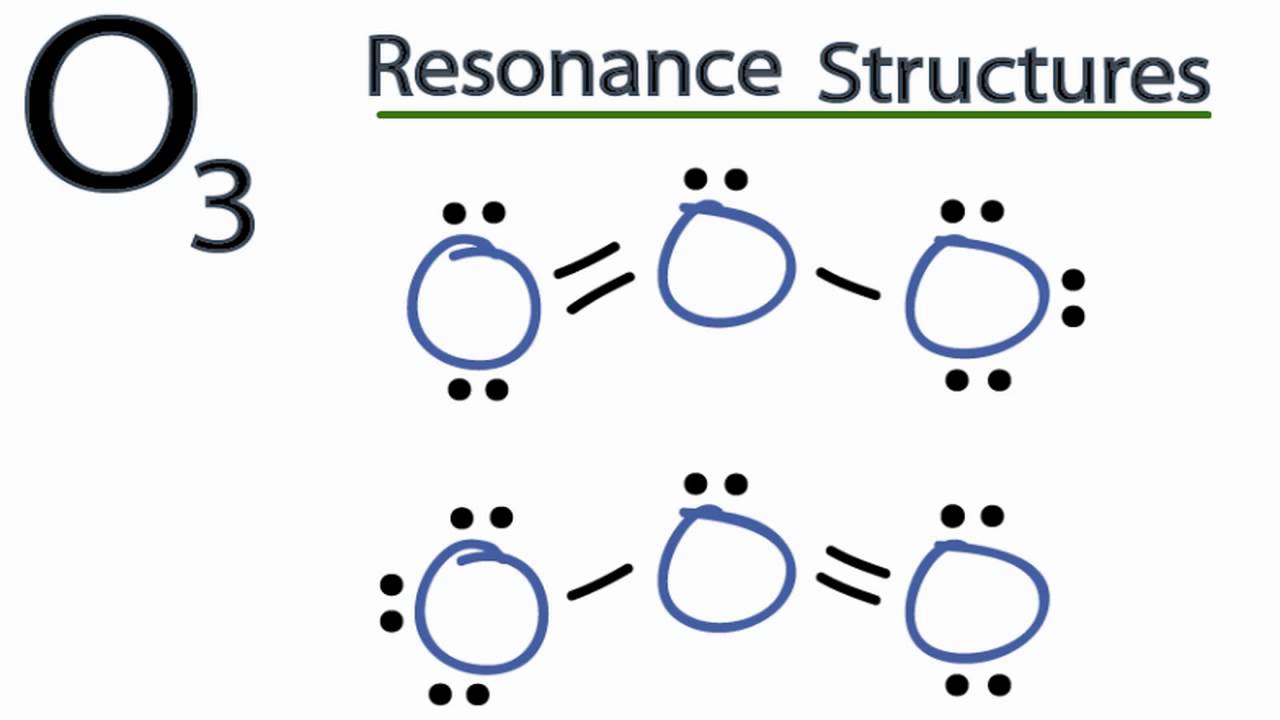
Explanation: A formal charge is equal to the number of valence electrons of an atom MINUS the number of electrons assigned to an atom. Consider the resonance structures for O3. Oxygen has 6 valence electrons. Look at the top left oxygen atom. It has two lone pairs ( 4 electrons) and a double bond ( 2 electrons).

Formal Charge 34. The formal charges on the three oxygen atoms in O3 mol..
Step 1. We divide the bonding electron pairs equally for all I-Cl bonds: Step 2. We assign lone pairs of electrons to their atoms. Each Cl atom now has seven electrons assigned to it, and the I atom has eight. Step 3. Subtract this number from the number of valence electrons for the neutral atom: I: 7 - 8 = -1.
O3 Lewis Structure Formal Charge Basics of Chemistry
Such is the case for ozone (\(\ce{O3}\)), an allotrope of oxygen with a V-shaped structure and an O-O-O angle of 117.5°. Ozone (\(O_3\)) 1. We know that ozone has a V-shaped structure, so one O atom is central:. We can convert each lone pair to a bonding electron pair, which gives each atom an octet of electrons and a formal charge of 0.
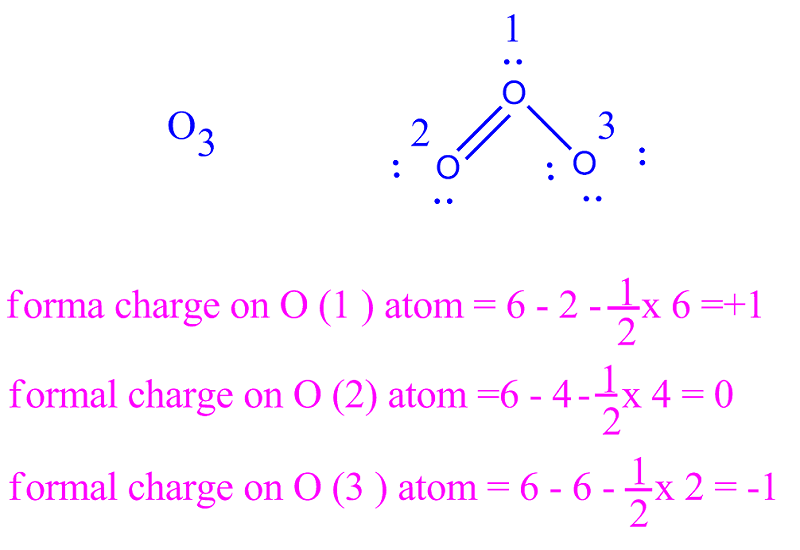
Formal charge, formal charge calculation and significance of formal charge PG.CHEMEASY
The formal charge of the ozone molecule is zero. Its Lewis structures do present charge separation. With simple VSEPR considerations, there are 18 valence electrons to distribute around the 3 oxygen atoms (24 electrons in total; 6 are inner core). Typically, a Lewis structure of O=stackrel(ddot)O^(+)-O^(-), would be depicted. Going from left O to right O and including the 2 inner core.

confusion over resonance major contributor and reactivity r/Mcat
Below is the formula for formal charge: Lewis Structure of O3. Here, we will be dealing with ozone, the molecular formula is O3. The below discussion, therefore, will be based on finding out the Lewis Structure of O3. Ozone consists of three oxygen atoms. Oxygen belongs to group VI of the periodic table with an atomic no of 8.
Solved Assign formal charges to each atom in the O3 molecule
Bonding electrons = 1 single bond = 2 electrons. Non-bonding electrons = 3 lone pairs = 3 (2) = 6 electrons. Formal charge on the single bonded Oxygen atom = 6 - 6 - 2/2 = 6 - 6 - 1 = 6 -7 = -1. ∴ The formal charge on the single-bonded O-atom in O3 is -1. This calculation shows that zero formal charges are present on double-bonded O.

O3 Lewis Structure Formal Charge Basics of Chemistry
How to Calculate the Formal charge of O3This video has answered the following questions:1)What is the formal charge of oxygen in ozone?2)Formal charge of O3.